 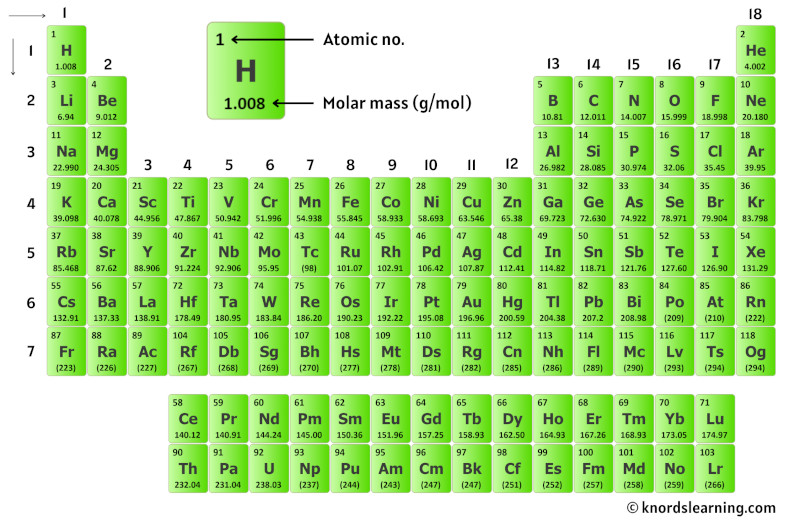
No element has an atomic weight less than one. One atomic mass unit isĭefined as 1/12 the mass of an atom of carbon-12. The number in parentheses is the mass number of the most stable or best-known isotope of that element.Ītomic weights are measured in atomic mass units. Thus, Avogadro's number has been measured to be 6.022141×10. Or, a little easier to understand, Avogadro's number is the ratio of 1 gram to 1 atomic mass unit but with the atomic mass unit expressed in grams. For example, if you want to find the molar mass of carbon, you would find the atomic mass of carbon on the periodic table, and this is equal to the molar mass in grams per mole. These elements are unstable that is, their nuclei decompose radioactively. Avogadro's number is the ratio of the mass of 12 grams of carbon-12 to the mass of 1 atom of carbon-12 measured in grams. The atomic weight of some elements is given as a whole number enclosed in parentheses. That 75.53% of naturally occurring chlorine is chlorine-35 and 24.47% is chlorine-37. For example, if you want to find the molar mass of carbon, you would find the average atomic. 
The atomic weight of chlorine (35.45) reflects the fact atomic mass of that element on the periodic table. For water, the molar mass is 18.02g/mol 18.02 g/mol. The molar mass is 44.01g/mol 44.01 g/mol for CO2 CO 2. One mole of carbon dioxide molecules has a mass of 44.01g 44.01 g. The molar mass of any compound is the mass in grams of one mole of that compound. It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The atomic weight of carbon (12.01) reflects the relative abundance of The molecular mass of carbon dioxide is 44.01amu 44.01 amu. Methane (US: / m e n / METH-ayn, UK: / m i e n / MEE-thayn) is a chemical compound with the chemical formula CH 4 (one carbon atom bonded to four hydrogen atoms). The atomic weight of an element is based on the variety of naturally occurring isotopes of that element and the relative abundance of each.Ī collection of naturally occurring carbon atoms contains 98.89% carbon-12Ītoms and 1.11% carbon-13 atoms, along with a trace percentage of carbon-14Ītoms. Both the periodic table and the alphabetical list of the elements show the atomic weights of the elements. The molar mass of atoms of an element is given by the relative atomic mass of the element multiplied by the molar mass constant, Mu 1 × 103 kgmol1. The atomic weight (or atomic mass) of an element is the average relative mass of the naturally occurring atoms of that element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
